TLDR: BigHat has demonstrated the first-known NOT-gated, T cell-engaging multispecific antibody, uniquely enabled through AI/ML-enabled structural and affinity engineering not possible with conventional antibody design. NOT gates enable efficient targeting of diseased cells while sparing healthy cells based on specific genetic or transcriptional patterns.
T cell engagers (TCEs) have transformed the treatment of devastating blood cancers by redirecting T cells to kill cancer cells. However, in solid tumors, the same potency that makes TCEs powerful can also limit their therapeutic potential. Even low-level expression of tumor-associated antigens (TAA) on healthy tissues can trigger remarkable on-target, off-tumor toxicity. There are very few tumor-specific and broadly expressed protein targets, presenting a substantial roadblock to the development of safe and effective TCEs. BigHat’s AI/ML models for protein engineering are designing the next generation of therapeutics that leverage readily detectable patterns of TAAs that uniquely decode cancer cells within healthy tissues.
Boolean logic gates—particularly A NOT (A+B) architectures—have emerged as a strategy to increase tumor selectivity. NOT gates are especially compelling: they encode an “off switch,” blocking T cell activity in the presence of an antigen found on healthy tissues (Normal Tissue-associated antigen, “nTAA”) while permitting activity where that antigen is absent. To date, this approach has been largely confined to cellular therapies using dual activating/inhibitory CAR (CAR/iCAR) designs. A recombinant antibody-based NOT-gated TCE combining logic control with the scalability and off-the-shelf advantages of protein therapeutics has not been demonstrated.
BigHat has achieved the world’s first demonstration of an antibody-based NOT-gated TCE and developed this technology into a plug-and-play platform built on a three-dimensional engineered antibody scaffold, dynamically antagonizing CD3 agonism in the presence of a given nTAA. In effect, the molecule computes A NOT (A+B) at the cell surface through spatial design.
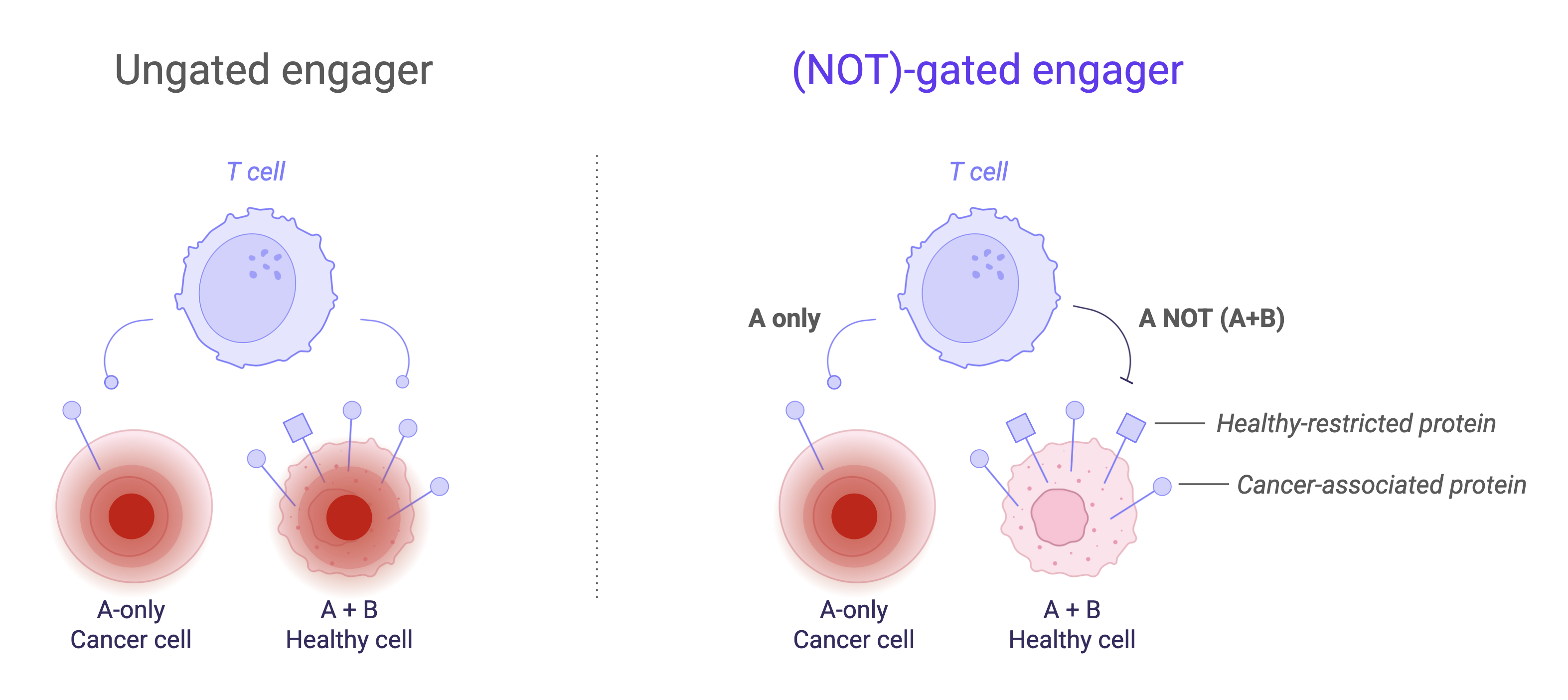
We began with a proof-of-concept system using Target X as a model safety antigen and CEACAM6 as the TAA. Trispecific designs (CD3, CEACAM6, X) demonstrated monovalent binding to all three antigens. The target X and CD3 paratopes are spatially highly approximated, facilitating (CD3 OR X) binding, but not both targets at the same time. In T cell-dependent cytotoxicity assays using primary T cells, this antibody leads to >50-fold specific lysis of GP2d colon cancer cells expressing CEACAM6-only as compared to GP2d cells expressing CEACAM6 and X. These data confirm that steric interference at the CD3 paratope can facilitate functional NOT-gated behavior.
We then systematically mapped the design space by manipulating and modeling the impact of linker length, antibody scaffold rigidity, and target binding affinities in successive design-build-test cycles. Consistently, a more rigid scaffold with a closer approximation of CD3 and X paratopes and lower CD3 affinity improved the NOT-gate selectivity. Real-time cell-killing assays support the mechanism, revealing accelerated and more complete cytotoxicity in X–negative target cells while preserving double-positive cells.

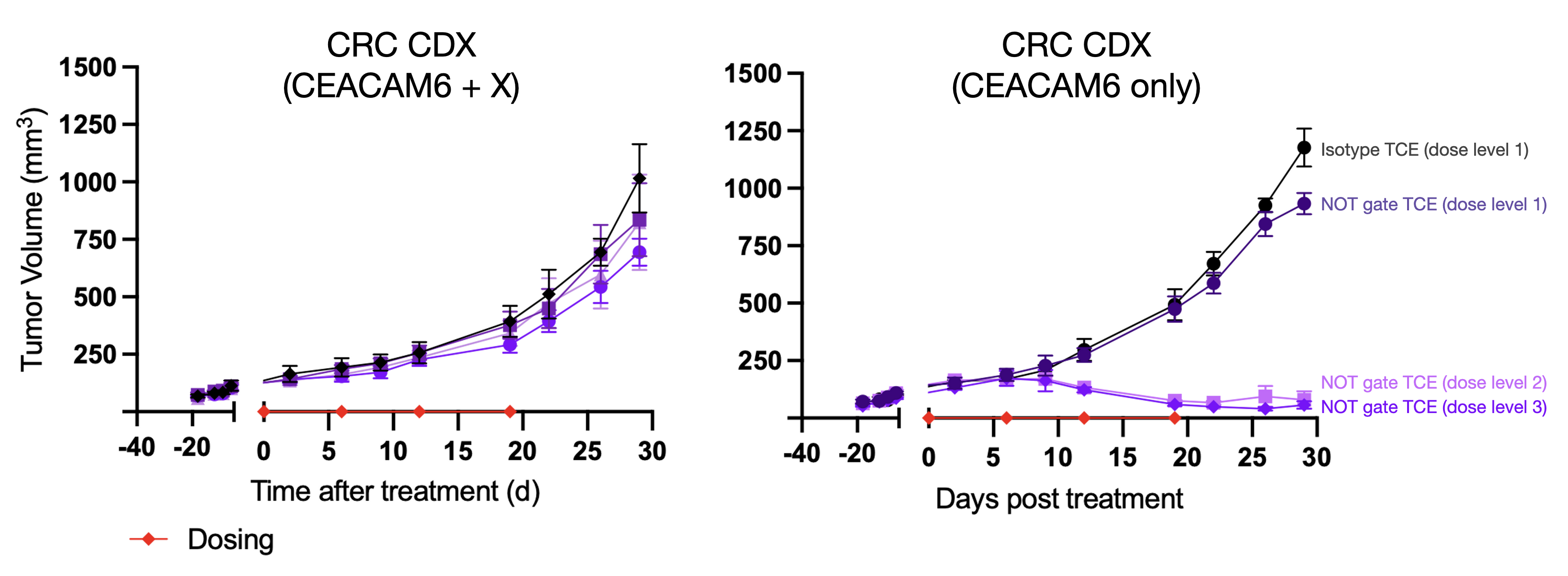
We also confirmed the NOT-gate functions similarly in vivo. In mice bearing CEACAM6+ xenografts, the lead molecule drove deep tumor regressions over multiple dose levels compared to a non-targeted TCE. In mice bearing isogenic X+ xenografts, no antitumor effects were observed over the same dose range.
To demonstrate modularity and clinical relevance, we extended the platform to a genetically defined oncology paradigm: HLA-A02 loss of heterozygosity (LOH). HLA LOH is a clonally fixed mechanism of immune escape present in a meaningful fraction of solid tumors. Using publicly characterized HLA-A02 binders as safety arms, we engineered trispecific NOT-gated TCEs that selectively activated T cells against HLA-A02–deficient, CEACAM5+ cancer cells while dampening activity in HLA-A02/CEACAM5 double-positive cells. With no optimization of affinities of the binders, we see about a 10-fold NOT gated function that can be readily optimized in subsequent design cycles.

Taken together, this work establishes that ML-guided design of TCEs can unlock novel therapeutic mechanisms, such as NOT logic, with extraordinary potential for patients through precise control over affinity, geometry, and scaffold architecture. NOT logic enables the targeting of cells based on intrinsic transcriptional or genomic features, such as loss of heterozygosity or indels, in oncology. NOT gates may also be useful for the safe and transformative treatment of diverse autoimmunities and latent viral infections through targeting unique features of pathological cells that are otherwise untargetable.
More broadly, this program illustrates how BigHat’s antibody engineering capabilities—spanning scaffold design, affinity tuning, biophysical optimization, and rapid functional screening—can uniquely unlock antibody-based modalities that were previously confined to cell therapy.









